Designing for Color blindness
Color choices and transformations for deuteranopia and other afflictions
Here, I help you understand color blindness and describe a process by which you can make good color choices when designing for accessibility.
The opposite of color blindness is seeing all the colors and I can help you find 1,000 (or more) maximally distinct colors.
You can also delve into the mathematics behind the color blindness simulations and learn about copunctal points (the invisible color!) and lines of confusion.
Color blindness R code
R code for converting an RGB color for color blindness. For details see the math tab and the resources section for background reading.
--- title: 'RGB color correction for color blindess: protanopia, deuteranopia, tritanopia' author: 'Martin Krzywinski' web: http://mkweb.bcgsc.ca/colorblind --- ```{r} gamma = 2.4 ############################################### # Linear RGB to XYZ # https://en.wikipedia.org/wiki/SRGB XYZ = matrix(c(0.4124564, 0.3575761, 0.1804375, 0.2126729, 0.7151522, 0.0721750, 0.0193339, 0.1191920, 0.9503041), byrow=TRUE,nrow=3) SA = matrix(c(0.2126,0.7152,0.0722, 0.2126,0.7152,0.0722, 0.2126,0.7152,0.0722),byrow=TRUE,nrow=3) ############################################### # XYZ to LMS, normalized to D65 # https://en.wikipedia.org/wiki/LMS_color_space # Hunt, Normalized to D65 LMSD65 = matrix(c( 0.4002, 0.7076, -0.0808, -0.2263, 1.1653, 0.0457, 0 , 0 , 0.9182), byrow=TRUE,nrow=3) # Hunt, equal-energy illuminants LMSEQ = matrix(c( 0.38971, 0.68898,-0.07868, -0.22981, 1.18340, 0.04641, 0 , 0 , 1 ), byrow=TRUE,nrow=3) # CIECAM97 SMSCAM97 = matrix(c( 0.8951, 0.2664, -0.1614, -0.7502, 1.7135, 0.0367, 0.0389, -0.0685, 1.0296), byrow=TRUE,nrow=3) # CIECAM02 LMSCAM02 = matrix(c( 0.7328, 0.4296, -0.1624, -0.7036, 1.6975, 0.0061, 0.0030, 0.0136, 0.9834), byrow=TRUE,nrow=3) ############################################### # Determine the color blindness correction in LMS space # under the condition that the correction does not # alter the appearance of white as well as # blue (for protanopia/deuteranopia) or red (for tritanopia). # For achromatopsia, greyscale conversion is applied # to the linear RGB values. getcorrection = function(LMS,type="p",g=gamma) { red = matrix(c(255,0,0),nrow=3) blue = matrix(c(0,0,255),nrow=3) white = matrix(c(255,255,255),nrow=3) LMSr = LMS %*% XYZ %*% apply(red,1:2,linearize,g) LMSb = LMS %*% XYZ %*% apply(blue,1:2,linearize,g) LMSw = LMS %*% XYZ %*% apply(white,1:2,linearize,g) if(type == "p") { x = matrix(c(LMSb[2,1],LMSb[3,1], LMSw[2,1],LMSw[3,1]),byrow=T,nrow=2) y = matrix(c(LMSb[1,1],LMSw[1,1]),nrow=2) ab = solve(x) %*% y C = matrix(c(0,ab[1,1],ab[2,1],0,1,0,0,0,1),byrow=T,nrow=3) } else if (type == "d") { x = matrix(c(LMSb[1,1],LMSb[3,1], LMSw[1,1],LMSw[3,1]),byrow=T,nrow=2) y = matrix(c(LMSb[2,1],LMSw[2,1]),nrow=2) ab = solve(x) %*% y C = matrix(c(1,0,0,ab[1,1],0,ab[2,1],0,0,1),byrow=T,nrow=3) } else if (type == "t") { x = matrix(c(LMSr[1,1],LMSr[2,1], LMSw[1,1],LMSw[2,1]),byrow=T,nrow=2) y = matrix(c(LMSr[3,1],LMSw[3,1]),nrow=2) ab = solve(x) %*% y C = matrix(c(1,0,0,0,1,0,ab[1,1],ab[2,1],0),byrow=T,nrow=3) } else if (type == "a" | type == "g") { C = matrix(c(0.2126,0.7152,0.0722, 0.2126,0.7152,0.0722, 0.2126,0.7152,0.0722),byrow=TRUE,nrow=3) } return(C) } # rgb is a column vector convertcolor = function(rgb,LMS=LMSD65,type="d",g=gamma) { C = getcorrection(LMS,type) if(type == "a" | type == "g") { T = SA } else { M = LMS %*% XYZ Minv = solve(M) T = Minv %*% C %*% M } print(T) rgb_converted = T %*% apply(rgb,1:2,linearize,g) return(apply(rgb_converted,1:2,delinearize,g)) } # This function implements the method by Vienot, Brettel, Mollon 1999. # The approach is the same, just the values are different. # http://vision.psychol.cam.ac.uk/jdmollon/papers/colourmaps.pdf convertcolor2 = function(rgb,type="d",g=2.2) { xyz = matrix(c(40.9568, 35.5041, 17.9167, 21.3389, 70.6743, 7.98680, 1.86297, 11.4620, 91.2367),byrow=T,nrow=3) lms = matrix(c(0.15514, 0.54312, -0.03286, -0.15514, 0.45684,0.03286, 0,0,0.01608),byrow=T,nrow=3) rgb = (rgb/255)**g if(type=="p") { S = matrix(c(0,2.02344,-2.52581,0,1,0,0,0,1),byrow=T,nrow=3) rgb = 0.992052*rgb+0.003974 } else if(type=="d") { S = matrix(c(1,0,0,0.494207,0,1.24827,0,0,1),byrow=T,nrow=3) rgb = 0.957237*rgb+0.0213814 } else { stop("Only type p,d defined for this function.") } M = lms %*% xyz T = solve(M) %*% S %*% M print(T) rgb = T %*% rgb rgb = 255*rgb**(1/g) return(rgb) } ############################################### # RGB to Lab rgb2lab = function(rgb,g=gamma) { rgb = apply(rgb,1:2,linearize,g) xyz = XYZ %*% rgb delta = 6/29 xyz = xyz / (c(95.0489,100,108.8840)/100) f = function(t) { if(t > delta**3) { return(t**(1/3)) } else { return (t/(3*delta**2) + 4/29) } } L = 116*f(xyz[2]) - 16 a = 500*(f(xyz[1]) - f(xyz[2])) b = 200*(f(xyz[2]) - f(xyz[3])) return(matrix(c(L,a,b),nrow=3)) } # CIE76 (https://en.wikipedia.org/wiki/Color_difference) deltaE = function(rgb1,rgb2) { lab1 = rgb2lab(rgb1) lab2 = rgb2lab(rgb2) return(sqrt(sum((lab1-lab2)**2))) } clip = function(v) { return(max(min(v,1),0)) } ############################################### # RGB to/from linear RGB #https://en.wikipedia.org/wiki/SRGB linearize = function(v,g=gamma) { if(v <= 0.04045) { return(v/255/12.92) } else { return(((v/255 + 0.055)/1.055)**g) } } delinearize = function(v,g=gamma) { if(v <= 0.003130805) { return(255*12.92*clip(v)) } else { return(255*clip(1.055*(clip(v)**(1/g))-0.055)) } } pretty = function(x) { noquote(formatC(x,digits=10,format="f",width=9)) } # a dark red rgb1 = matrix(c(0,209,253),nrow=3) # dark green rgb2 = matrix(c(60,135,0),nrow=3) # simulate deuteranopia convertcolor(rgb1,type="d") convertcolor(rgb2,type="d") # get color distance before and after simulation deltaE(rgb1,rgb2) deltaE(convertcolor(rgb1,type="d"),convertcolor(rgb2,type="d")) # transformation matrices for each color blindness type M = LMSD65 %*% XYZ pretty(solve(M) %*% getcorrection(LMSD65,"p") %*% M) pretty(solve(M) %*% getcorrection(LMSD65,"d") %*% M) pretty(solve(M) %*% getcorrection(LMSD65,"t") %*% M) pretty(SA) # method by Vienot, Brettel, Mollon, 1999 convertcolor2(rgb1,type="d",g=2.2) convertcolor2(rgb2,type="d",g=2.2) ```
# a dark red rgb1 = matrix(c(225,0,30),nrow=3) # dark green rgb2 = matrix(c(60,135,0),nrow=3) # simulate deuteranopia convertcolor(rgb1,type="d") [,1] [1,] 136.7002 [2,] 136.7002 [3,] 0.0000 convertcolor(rgb2,type="d") [,1] [1,] 116.76071 [2,] 116.76071 [3,] 16.73263 # get color distance before and after simulation deltaE(rgb1,rgb2) [1] 116.9496 deltaE(convertcolor(rgb1,type="d"),convertcolor(rgb2,type="d")) [1] 12.72204 # transformation matrices for each color blindness type M = LMSD65 %*% XYZ pretty(solve(M) %*% getcorrection(LMSD65,"p") %*% M) [,1] [,2] [,3] [1,] 0.1705569911 0.8294430089 0.0000000000 [2,] 0.1705569911 0.8294430089 -0.0000000000 [3,] -0.0045171442 0.0045171442 1.0000000000 pretty(solve(M) %*% getcorrection(LMSD65,"d") %*% M) [,1] [,2] [,3] [1,] 0.3306600735 0.6693399265 -0.0000000000 [2,] 0.3306600735 0.6693399265 0.0000000000 [3,] -0.0278553826 0.0278553826 1.0000000000 pretty(solve(M) %*% getcorrection(LMSD65,"t") %*% M) [,1] [,2] [,3] [1,] 1.0000000000 0.1273988634 -0.1273988634 [2,] -0.0000000000 0.8739092990 0.1260907010 [3,] 0.0000000000 0.8739092990 0.1260907010 pretty(SA) [,1] [,2] [,3] [1,] 0.2126000000 0.7152000000 0.0722000000 [2,] 0.2126000000 0.7152000000 0.0722000000 [3,] 0.2126000000 0.7152000000 0.0722000000 # method by Vienot, Brettel, Mollon, 1999 convertcolor2(rgb1,type="d",g=2.2) [,1] [,2] [,3] [1,] 0.29275003 0.70724967 -2.978356e-08 [2,] 0.29275015 0.70724997 1.232823e-08 [3,] -0.02233659 0.02233658 1.000000e+00 [,1] [1,] 131.81223 [2,] 131.81226 [3,] 36.37274 convertcolor2(rgb2,type="d",g=2.2) [,1] [,2] [,3] [1,] 0.29275003 0.70724967 -2.978356e-08 [2,] 0.29275015 0.70724997 1.232823e-08 [3,] -0.02233659 0.02233658 1.000000e+00 [,1] [1,] 122.71798 [2,] 122.71801 [3,] 48.34316
Nasa to send our human genome discs to the Moon
We'd like to say a ‘cosmic hello’: mathematics, culture, palaeontology, art and science, and ... human genomes.



Comparing classifier performance with baselines
All animals are equal, but some animals are more equal than others. —George Orwell
This month, we will illustrate the importance of establishing a baseline performance level.
Baselines are typically generated independently for each dataset using very simple models. Their role is to set the minimum level of acceptable performance and help with comparing relative improvements in performance of other models.
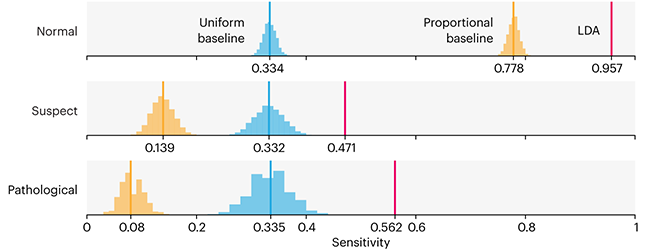
Unfortunately, baselines are often overlooked and, in the presence of a class imbalance5, must be established with care.
Megahed, F.M, Chen, Y-J., Jones-Farmer, A., Rigdon, S.E., Krzywinski, M. & Altman, N. (2024) Points of significance: Comparing classifier performance with baselines. Nat. Methods 20.
Happy 2024 π Day—
sunflowers ho!
Celebrate π Day (March 14th) and dig into the digit garden. Let's grow something.
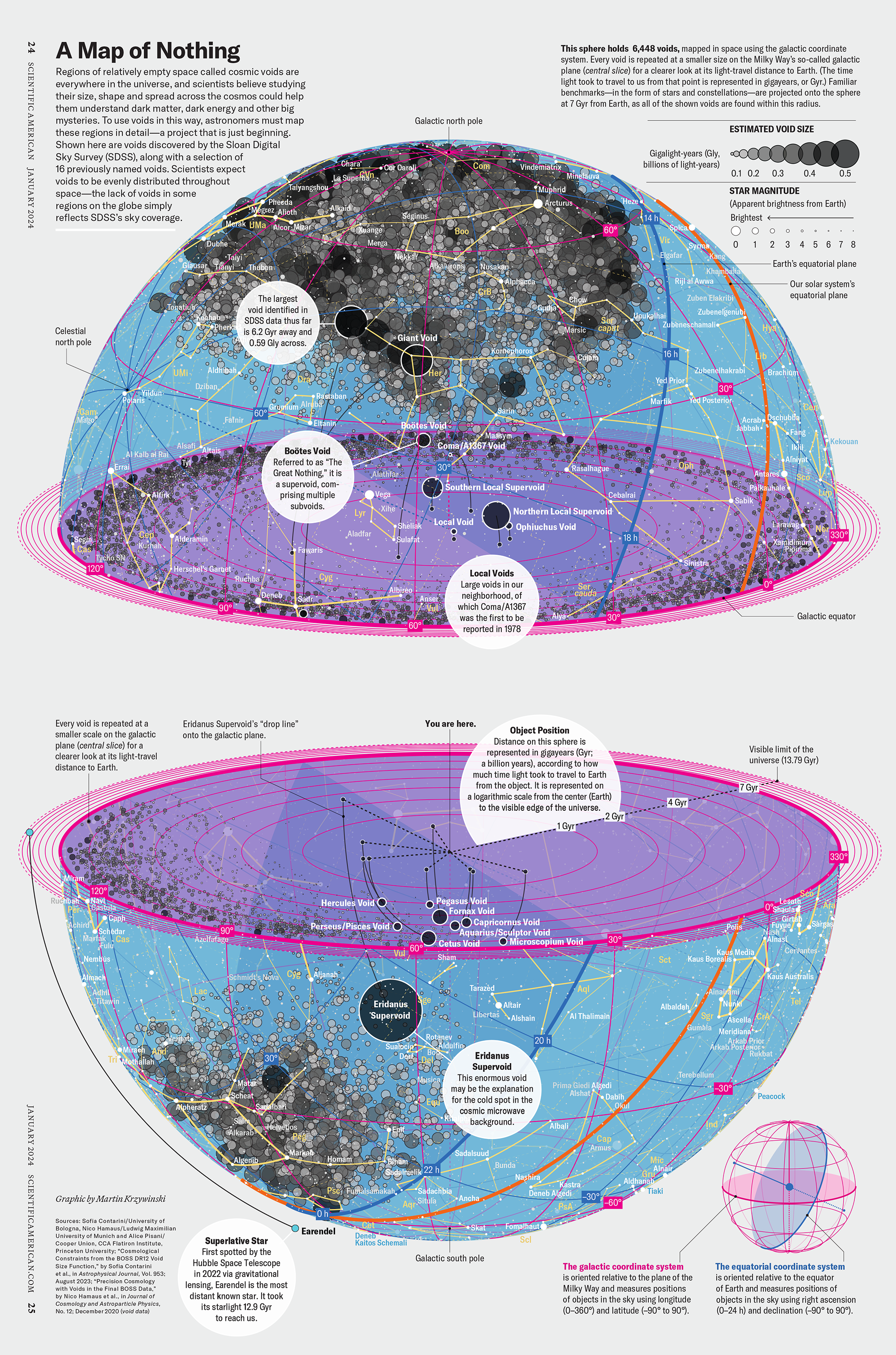
How Analyzing Cosmic Nothing Might Explain Everything
Huge empty areas of the universe called voids could help solve the greatest mysteries in the cosmos.
My graphic accompanying How Analyzing Cosmic Nothing Might Explain Everything in the January 2024 issue of Scientific American depicts the entire Universe in a two-page spread — full of nothing.
The graphic uses the latest data from SDSS 12 and is an update to my Superclusters and Voids poster.
Michael Lemonick (editor) explains on the graphic:
“Regions of relatively empty space called cosmic voids are everywhere in the universe, and scientists believe studying their size, shape and spread across the cosmos could help them understand dark matter, dark energy and other big mysteries.
To use voids in this way, astronomers must map these regions in detail—a project that is just beginning.
Shown here are voids discovered by the Sloan Digital Sky Survey (SDSS), along with a selection of 16 previously named voids. Scientists expect voids to be evenly distributed throughout space—the lack of voids in some regions on the globe simply reflects SDSS’s sky coverage.”
voids
Sofia Contarini, Alice Pisani, Nico Hamaus, Federico Marulli Lauro Moscardini & Marco Baldi (2023) Cosmological Constraints from the BOSS DR12 Void Size Function Astrophysical Journal 953:46.
Nico Hamaus, Alice Pisani, Jin-Ah Choi, Guilhem Lavaux, Benjamin D. Wandelt & Jochen Weller (2020) Journal of Cosmology and Astroparticle Physics 2020:023.
Sloan Digital Sky Survey Data Release 12
Alan MacRobert (Sky & Telescope), Paulina Rowicka/Martin Krzywinski (revisions & Microscopium)
Hoffleit & Warren Jr. (1991) The Bright Star Catalog, 5th Revised Edition (Preliminary Version).
H0 = 67.4 km/(Mpc·s), Ωm = 0.315, Ωv = 0.685. Planck collaboration Planck 2018 results. VI. Cosmological parameters (2018).
constellation figures
stars
cosmology
Error in predictor variables
It is the mark of an educated mind to rest satisfied with the degree of precision that the nature of the subject admits and not to seek exactness where only an approximation is possible. —Aristotle
In regression, the predictors are (typically) assumed to have known values that are measured without error.
Practically, however, predictors are often measured with error. This has a profound (but predictable) effect on the estimates of relationships among variables – the so-called “error in variables” problem.
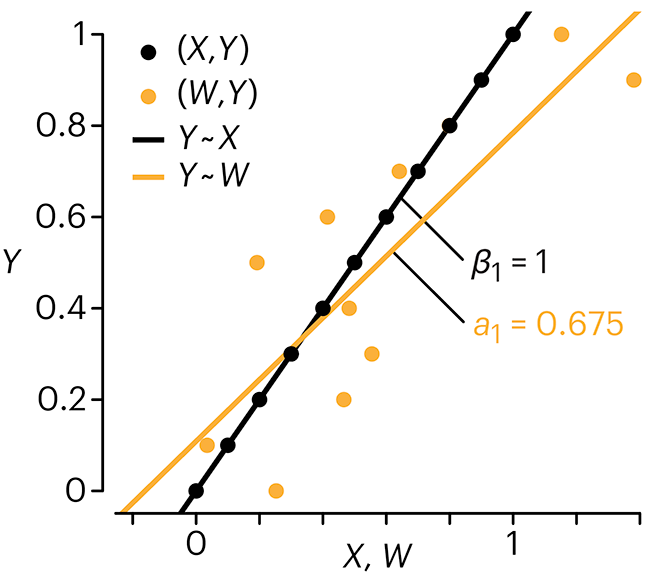
Error in measuring the predictors is often ignored. In this column, we discuss when ignoring this error is harmless and when it can lead to large bias that can leads us to miss important effects.
Altman, N. & Krzywinski, M. (2024) Points of significance: Error in predictor variables. Nat. Methods 20.
Background reading
Altman, N. & Krzywinski, M. (2015) Points of significance: Simple linear regression. Nat. Methods 12:999–1000.
Lever, J., Krzywinski, M. & Altman, N. (2016) Points of significance: Logistic regression. Nat. Methods 13:541–542 (2016).
Das, K., Krzywinski, M. & Altman, N. (2019) Points of significance: Quantile regression. Nat. Methods 16:451–452.